User Guide –
Before Beamtime
Things You Need to Do
Before arriving at CLS for your scheduled beamtime, please ensure you have updated you project to meet the following criteria:
- SAMPLES/MATERIALS/EQUIPMENT
- Any samples/materials/equipment that you plan on bringing must be listed on the project
- Without prior approval your samples/materials will not be permitted to be used at CLS
- You must provide an MSDS for all materials/samples identified as hazardous
- If you are planning to use CLS equipment, click here for a list of available equipment. Please note that some equipment requires pre-booking. Please contact your beamline support for booking assistance.
- To amend samples/materials/equipment, click the “Amend” icon under the “Samples & Materials” section on the project page.
- NOTE: Amendments should be submitted a minimum of 2 weeks before your first shift to allow sufficient time for safety and technical reviews to occur. CLS cannot guarantee that amendments received outside of the 2-week period will be approved before your beamtime.
- TEAM MEMBERS
- All team members participating in the upcoming experiment must have the appropriate training and agreements (User and Institutional) completed. Complete training online at https://training.lightsource.ca/
- To edit your team members, click the “Edit Team” icon under the “Team Members” section on the project page.
- Participating team members must meet the requirements for obtaining a facility access badge.
Beamline Specific Orientation (BSO)
In addition to CLS online training, all BMIT users that will be present for experiment are required to attend an in-person Beamline Specific Orientation (BSO). Your local contact will arrange the BSO for you. The BSO will cover the beamline specific rules, policies, and safety points. If you like you can read a copy of the BSO in advance.
Get in Touch with Your Local Contact
Each experiment at BMIT is supported by a BMIT scientist that will be your primary point of contact for all inquiries about your beamtime. Your contact will reach you several weeks prior to the start of your beamtime in order to discuss details of your experiment. We encourage our users to contact BMIT staff well in advance if they have questions about their experimental setup, samples, data processing, etc. Any major changes to the proposed experiment needs to be discussed directly with BMIT Beamline Responsible in advance and there will be no guarantee that it will be accepted.
Samples
Sample Shape and Size
In general, the ideal sample for tomography is a cylindrical sample that is not extremely absorbing. BMIT can deliver up to 150keV X-rays, however, the photon flux will be low at high energies. In addition, the absorption cross section of all elements decreases as the incident photon energy increases. For highly absorbing samples, the sample should be cut to smaller sections, if that is an option. Samples with very few absorbing features and generally uniform attenuation coefficient MUST be smaller than field of view of the detector. While most of samples scanned at BMIT range from a few mm to few cm, BMIT can accommodate larger samples with thickness in several tens of cm, however, if you have a large sample it is strongly advised to contact BMIT staff to look at the feasibility of your experiment before submitting a proposal.
Biological Materials and Live Animals
If you will be imaging biological or biohazardous samples please consult with CLS Biosafety document.
If you will be imaging live animals, please read the CLS Animal Care and Use document.
Shipping of Samples and Equipment
Please consult the CLS policy on shipping.
Ethics
All research performed at the CLS that involves: human participants and/or research using human biological materials, derived from living and deceased individuals, or live animals and/or animal tissue or biological fluids from animals requires ethics approval prior to performing research at the CLS. For a detailed description of the types of samples and experiments that require ethics approval please read the CLS ethics policy.
Sample Holder
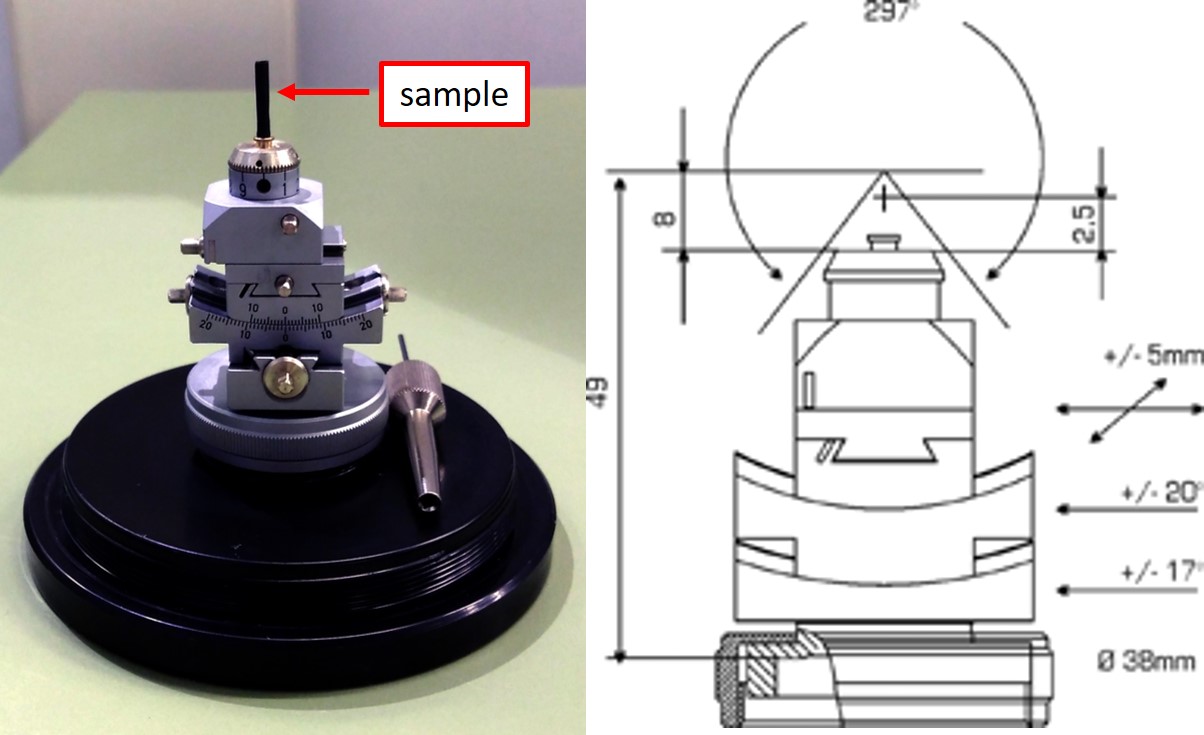
BMIT has an array of detectors with field of view from 3mm to 240mm in width that correspond to pixel sizes from 1.4um to 200um. Having a good and stable sample holder is very essential for collecting a good tomography data. In general, sample is placed in a sample holder (obtained by user) and then mounted on the specific rotation stage (BMIT equipment). When designing or purchasing a sample holder, attention should be paid to the bolt pattern of the rotary stages. You can find the images and drawing of our rotary stages below. Alternatively, many small samples as well as irregular samples can be held by a universal sample holder with no need to bolting them to the rotary stage.
High resolution micro-CT experiments require precise positioning and alignment of the sample in the beam. For those experiments, samples are pre-aligned on a pin using a goniometer. In-vivo experiments often require custom-made sample holders depending on the animal species and the area to be imaged. While obtaining all animal holders are the responsibility of the research groups, BMIT staff may assist you in designing and testing of the holders. If you require such assistance please contact your primary point of contact in advance. It is strongly recommended to discuss any experimental details with staff before preparing any sample holders or devices that are intended to be used in an experiment.
Universal Sample Holder
Drawing and additional information are available.
micro-CT Sample Holder
Drawing and additional information are available: model 1002 and model 1005.
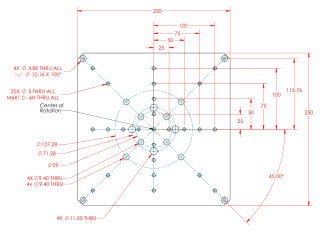
Kohzu RA16A-W Rotation Stage
The first Kohzu stage is a Kohzu RA16A-W rotation stage. Drawings and additional information are available.
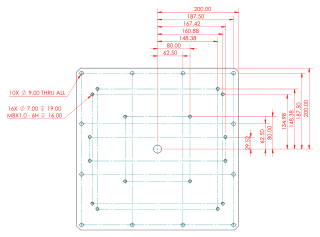
Kohzu RA16A-WH Rotation Stage

The second Kohzu stage is a Kohzu RA16A-WH rotation stage. Drawings and additional information are available.
05ID-2 CT Mounting Plate
This is a drawing of the hole pattern for a 05ID-2 CT mounting plate. Available as PDF.
MRT Lift Top Plate
This is a drawing of the hole pattern for a MRT Lift Top Plate. Available as PDF.
Thorlabs KB75/M Kinematic Base
One mounting option is a Thorlabs KB75/M Kinematic Base with M6 mounting. Auto CAD drawing is available.
Thorlabs NR360 S/M Rotation Stage
One rotation stage is the Thorlabs NR360S/M with SM2-Threaded Center Hole, Metric. Auto CAD drawing is available.
Access to Labs
BMIT has two labs designed to contain, house and prepare samples and live animals for live imaging. These labs also offer direct access to the experimental hutches. Basic laboratory equipment is available in the labs, including:
- Animal containment area
- Change room (with shower)
- Refrigerator/-20℃ freezer
- -80℃ freezer
- Biosafety cabinet and fume hood
- Vacuum desiccator
- Anesthesia gas system
- Warming oven
- Microwave
- Ultrasonic cleaner
Small Animal Lab


Large Animal Lab

In addition to BMIT labs, BMIT users can access other labs throughout the CLS. Please note that a valid permit and in advance booking of labs other than BMIT labs are required. For more details on types of equipment available and details on booking those labs please visit here.